Organic Chemistry Woodward-Hoffmann rules
Question
Compund A undergoes a cascade of electrocyclic reactions to give compound B under thermal conditions, and C under photochemical conditions. Compounds B and C can undergo a further cycloaddition reaction under photochemical conditions to give compounds D and E, respectively. Please write the chemical structures of compounds
B, C, D, and E. For the cascade of your choice (A-B-D or A-C-E), explain in detail the Il reaction mechanism. Considering the Woodward-Hoffmann rules and frontier molecular
Answer
Concept Introduction:
Woodward–Hoffmann rules:
Woodward–Hoffmann gives some rules, which are used to predict the stereochemistry of the products of electrocyclic reactions.
They are,
These rules are based on number of pi electrons present in the compound and based on conditions (light or heat), the orbital motion of a compound results the various stereochemistry products,
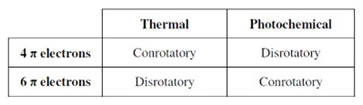
A system undergo conrotatory, to give trans product
A system undergo disrotatory, to give cis product
Answer:
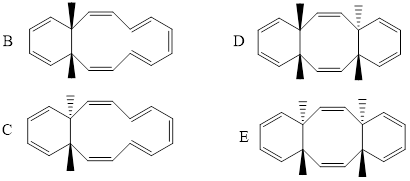
Explanation:
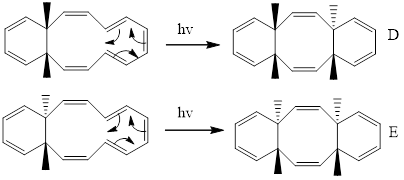
Given reaction is
,
The given compound undergoes 6 pi electron cyclization reactions.
According to Woodward-hoffmann rule, 6 pi electron systems gives conrotatory product under light and disrotatory under heat.
Therefore, the given compound gives cis product under heat condition and trans product under light condition.
The precuts are,
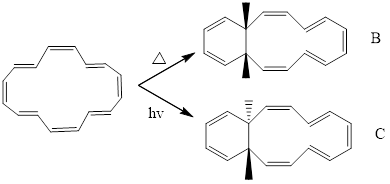
After the above cyclization, another part of 6 pi electron system undergoes cyclization under the condition of light.
In this cyclization takes place via conrotatory motion so trans products are formed.
Conclusion:
Products of given reaction are predicted by using Woodward-hoffmann rules.












